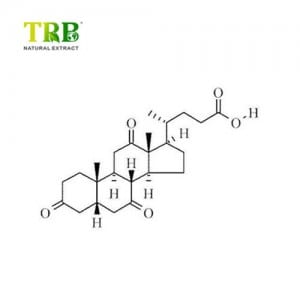
Acidum deoxycholicum (deoxycholatum basi coniugatum), etiam ut acidum cholanoicum et 3α, 12α-dihydroxy-β-cholan-24-oic acidum notum est, acidum bile est.
Acidum deoxycholicum unum est ex acidis bilis secundariis, quae sunt metabolicae per productiones bacterii intestinalis.Duo acida prima bilis ab hepate secreta sunt Acidum cholicum et Acidum chenodeoxycholicum.Bacteria metabolizant acidum chenodeoxycholicum in acidum bile secundarium acidum lithocholicum, et metaboliscunt acidum cholicum in acidum deoxycholicum.Accedunt secundae bilis acida, ut acidum ursodeoxycholicum.Acidum deoxycholicum in alcohole et acetico solutum est.Cum pura, fit in albo pulveris crystallini coloris ad formam.
Product nomen:Dehydrocholic acidum
CAS No: 81-23-2
Min: 98.0% Min
Color: White ad off-alba cum odore et sapore proprium
GMO Status: GMO Free
Stipare: in 25kgs fibra tympana
Repono: Contine vas pervium in loco frigido, loco sicco, prohibe a luce firma
Shelf Life: 24 months from the date of production
Officium
(1) Efficax est in multis generibus dapibus commercii perturbandi et dissociandi
(2) Usus sodii deoxycholati emergens est sicut biologicum purgat ad cellulas lysas et cellularum et membranarum solubilis.
(3) Usurpatur in praeparatione et formula quorundam instrumentorum microbiologicorum diagnostica.
(4.) Utilis ad elutionem seu regenerationem quarundam affinitatis columnarum species.
Applicationem
(1) Adhibetur in emulsificatione adipum pro effusio intestini.Extra corpus in experimentis cholagogis adhibetur et in usu etiam est ad fel fel praecavendum et dissolvendum.
(2) Natrium deoxycholatum, sal sodium acidi deoxycholici, saepe pro biologico purgat ad cellulas lysas et ad partes cellularum et solubilisarum membranarum.
| Plura de TRB | ||
| Regulation certificatione | ||
| USFDA, CEP, KOSHER HALAL GMP ISO Testimonia | ||
| Reliable Quality | ||
| Prope XX annos, exportare 40 regiones et regiones, plusquam 2000 batchiae quae ab TRB productae sunt nullas habent quaestiones qualitates, singulare purificationis processum, immunditiae et castitatis imperium conveniunt USP, EP et CP | ||
| Comprehensive Quality System | ||
|
| Quality Assurance System | √ |
| Document imperium | √ | |
| Validation System | √ | |
| Training System | √ | |
| Internum Audit Protocollum | √ | |
| Supple Audit System | √ | |
| Equipment facilities Ratio | √ | |
| Material Control System | √ | |
| Productio Imperium System | √ | |
| Packaging Labeling System | √ | |
| Laboratorium Control System | √ | |
| Verificationis Validation System | √ | |
| Regulatory Negotiis Ratio | √ | |
| Imperium Totum Fontes et Processus | ||
| Materiam omnem rudis, accessiones et materias sarcinas stricte moderatas. Praeferrem materias crudas et accessiones et sarcinas cum US DMF numero. Multae materiae rudis praebitorum copia certificatur. | ||
| Fortis Cooperantem Institutiones sustentare | ||
| Institutum botanicum/Institutum microbiologiae/Academiae Scientiarum et Technologiae/Universitatis | ||