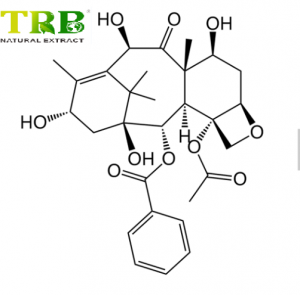
Product Name:Ursodeoxycholic acid Powder
Other Name:Bulk Ursodeoxycholic acid powder (UDCA),Ursodiol; UDCA; (3a,5b,7b,8x)-3,7-dihydroxycholan-24-oic acid; Ursofalk; Actigall; Urso
CAS No:128-13-2
Assay: 99%~101%
Colour: Off White to Pale Yellow Powder
Solubility:soluble in water, freely soluble in ethyl alcohol
GMO Status:GMO Free
Packing: in 25kgs fiber drums
Storage:Keep container unopened in cool, dry place,Keep away from strong light
Shelf Life:24 months from date of production
Ursodeoxycholic Acid Powder (CAS 128-13-2) – High-Purity Pharmaceutical and Research Grade
Product Overview
Ursodeoxycholic Acid (UDCA), also known as Ursodiol, is a hydrophilic secondary bile acid with a purity of ≥99% . It is synthesized from cholic acid through advanced chemical and enzymatic methods, ensuring high stereoselectivity and environmental sustainability . This white crystalline powder is widely used in pharmaceutical research and therapeutic applications due to its cytoprotective, anti-apoptotic, and cholesterol-lowering properties .
Key Specifications
- CAS No.: 128-13-2
- Molecular Formula: C₂₄H₄₀O₄
- Molecular Weight: 392.57 g/mol
- Melting Point: 203–204°C
- Storage: Store at -20°C in a dry, dark environment .
Mechanism of Action
UDCA exerts multiple therapeutic effects:
- Liver Protection: Enhances bile flow, reduces cholesterol saturation in bile, and stabilizes hepatocyte membranes .
- FATP5 Inhibition: Acts as a liver-specific fatty acid transport protein 5 (FATP5) inhibitor, modulating lipid metabolism .
- Immunomodulation: Suppresses oxidative stress and inflammatory pathways (e.g., NF-κB, Erk) .
- Gallstone Dissolution: Lowers cholesterol absorption and dissolves cholesterol gallstones via bile acid replacement .
Applications
1. Liver Diseases
- Primary Biliary Cholangitis (PBC): Improves liver enzyme levels (e.g., alkaline phosphatase) and delays disease progression .
- Non-Alcoholic Fatty Liver Disease (NAFLD): Reduces hepatic lipid accumulation .
- Cholestatic Disorders: Safely used in intrahepatic cholestasis of pregnancy at doses of 10–25 mg/kg/day .
2. Gallstone Management
- Effective for cholesterol gallstones in patients unsuitable for surgery .
3. Emerging Research
- COVID-19: Potential role in reducing ACE2 receptor expression, inhibiting viral entry .
- Cardioprotection: Investigated for anti-arrhythmic and anti-fibrotic effects .
Safety and Handling
- Hazard Statements: Causes eye and skin irritation (Category 2) .
- Precautions:Regulatory Status: For research use only. Not approved for human or veterinary therapeutic use .
- Use personal protective equipment (gloves, goggles).
- Avoid inhalation or direct contact.
Packaging and Ordering
- Available Quantities: 1 g, 10 g, 100 g.
- Custom Packaging: Bulk orders available upon request.
- COA Access: Batch-specific certificates provided with each order .
Why Choose Our UDCA Powder?
- High Purity: Rigorous QC ensures ≥99% purity via HPLC analysis .
- Research Support: Technical documentation and synthesis protocols available .
- Global Compliance: Meets ISO and GMP standards for pharmaceutical intermediates.
Keywords: Ursodeoxycholic Acid Powder, CAS 128-13-2, FATP5 Inhibitor, Cholesterol Gallstones, Liver Protection, ≥99% Purity