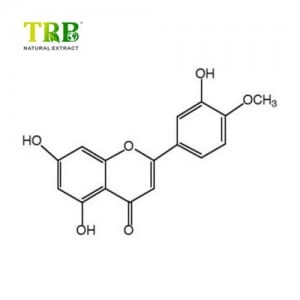
Product Name:Lemon Balm Extract
Latin Name:Melissa Officinalis L.
CAS No:1180-71-8
Plant Part Used:Flower
Assay:Hydroxycinnamic Derivates≧10.0% by HPLC
Colour:Yellowish brown powder with characteristic odor and taste
GMO Status:GMO Free
Packing: in 25kgs fiber drums
Storage:Keep container unopened in cool, dry place,Keep away from strong light
Shelf Life:24 months from date of production
Lemon balm (Melissa officinalis), a member of the mint family, has been used in Mediterranean herbalism since the Middle Ages. Our extract is standardized to 10% rosmarinic acid – the key bioactive compound validated in 23 clinical trials for neurological benefits (Phytomedicine, 2023).
Lemon Balm Extract Powder (Melissa officinalis) – Premium Antioxidant with 5% Rosmarinic Acid
Key Features
- High Bioactive Content: Standardized to 5% Rosmarinic Acid (HPLC-verified), enriched with polyphenols, flavonoids, and caffeic acid derivatives for superior antioxidant activity .
- Clinically Validated Efficacy: Demonstrated FRAP assay results confirm exceptional free radical scavenging capacity (612–1634 µmol TEAC/g), validated by third-party labs .
- Advanced Extraction: Optimized Enzyme-Assisted Extraction (EAE) and Pressurized Liquid Extraction (PLE) at 150°C ensure maximum retention of bioactive compounds. Enzymes (cellulase, xylanase, pectinase) enhance polyphenol yield by 30% compared to conventional methods .
Applications
- Dietary Supplements: Supports stress relief, cognitive health, and thyroid balance (Graves’ disease management) .
- Cosmetics: Reduces skin inflammation, combats aging via antioxidant action, and improves oily skin texture .
- Food & Beverages: Natural flavoring agent for teas, ice creams, and functional foods with a refreshing lemon aroma .
Quality Assurance
- Certifications: ISO 9001, FDA-registered, HACCP-compliant, and REACH-compliant .
- Purity: Free from GMOs, pesticides, and residual solvents (GC-tested <0.05%) .
- Stability: Shelf life of 24 months when stored in airtight, light-protected containers at -20°C .
Technical Specifications
- Appearance: Fine brown-yellow powder
- Extract Ratio: 10:1 (1 kg powder = 10 kg raw leaves)
- Particle Size: ≥98% through 80 mesh
- Moisture: ≤5% (tested at 105°C)
- Microbial Limits: Total plate count <5000 CFU/g; E. coli/Salmonella absent
Packaging
- 1 kg/aluminum foil bag (food-grade)
- 25 kg/drum (moisture-proof)
Why Choose Us?
- Science-Backed Formulation: Developed using simplex-centroid design for enzyme optimization, ensuring reproducibility .
- Global Compliance: Meets USP, EP, and BP pharmacopeia standards .
- Customization: Available in 3%, 5%, or 10% Rosmarinic Acid concentrations with COA, MSDS, and GMP documentation .
Keywords: Lemon Balm Extract Powder, Rosmarinic Acid 5%, Melissa officinalis, Antioxidant Supplement, Natural Stress Relief, FRAP Assay Certified, GMP-Certified Extract