Our company since its inception, usually regards product high-quality as company life, constantly boost manufacturing technology, boost product excellent and continuously strengthen company total excellent administration, in strict accordance using the national standard ISO 9001:2000 for Green Coffee Bean Extract, We have been willing to offer you the ideal strategies within the designs of the orders in a specialist way should you need. While in the meantime, we retain on acquiring new technologies and producing new designs so as to produce you ahead from the line of this small business.
Our company since its inception, usually regards product high-quality as company life, constantly boost manufacturing technology, boost product excellent and continuously strengthen company total excellent administration, in strict accordance using the national standard ISO 9001:2000 for China Coffea Arabica, Chlorogenic Acid, Our company, is always regarding quality as company’ s foundation, seeking for development via high degree of credibility , abiding by iso9000 quality management standard strictly , creating top-ranking company by spirit of progress-marking honesty and optimism.
Green coffee bean extract is made from the unroasted green beans of Coffea arabica L, whose nutrients have not been destroyed
and the nutritional value is higher than roasted coffee. Green coffee bean has strong anti-oxidant and fat accumulation
suppressing properties. The extract contains numerous polyphenolic compounds such as chlorogenic acid. These compounds
have been discovered to possess powerful antioxidant properties. Unroasted green coffee beans are a much better source
of antioxidants than roasted coffee beans.
Product Name:Green Coffee Bean Extract
Latin Name:Coffea Robusta/Coffea Arabica L.
CAS No:327-97-9
Plant Part Used:Seed
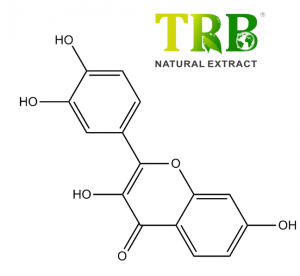
Assay:Chlorogenic Acids≧50.0% by HPLC
Colour: Brown fine powder with characteristic odor and taste
GMO Status:GMO Free
Packing: in 25kgs fiber drums
Storage:Keep container unopened in cool, dry place,Keep away from strong light
Shelf Life:24 months from date of production
Function:
-Chlorogenic acid, long known as an antioxidant with potential anti-cancer activity, also slows the release of glucose into bloodstream after a meal.
-lower one’sblood sugar level, suppress the appetite, lower blood pressure, and reduce levels of visceral fat.
-Useful in fighting the free radicals in our bodies that can damage our cells and contribute to conditions such as cardiovascular disease. Test results showed Green Coffee Bean had more than double the rate of oxygen radical absorbance capability when compared to green tea and grape seed extracts.
-Act as an effective painkiller especially for migraine medications;
-Reduce the risk of diabetes.
Application
-Food field, the coffee are most popular food among people;
-Pharmaceutical field, to be used as raw materials to reduce blood pressure;
-Cosmetic field, green bean oil can effectively regulate the skin, stimulate vigor and arouse the spirit.
TECHNICAL DATA SHEET
| Item | Specification | Method | Result |
| Identification | Positive Reaction | N/A | Complies |
| Extract Solvents | Water/Ethanol | N/A | Complies |
| Particle size | 100% pass 80 mesh | USP/Ph.Eur | Complies |
| Bulk density | 0.45 ~ 0.65 g/ml | USP/Ph.Eur | Complies |
| Loss on drying | ≤5.0% | USP/Ph.Eur | Complies |
| Sulphated Ash | ≤5.0% | USP/Ph.Eur | Complies |
| Lead(Pb) | ≤1.0mg/kg | USP/Ph.Eur | Complies |
| Arsenic(As) | ≤1.0mg/kg | USP/Ph.Eur | Complies |
| Cadmium(Cd) | ≤1.0mg/kg | USP/Ph.Eur | Complies |
| Solvents Residue | USP/Ph.Eur | USP/Ph.Eur | Complies |
| Pesticides Residue | Negative | USP/Ph.Eur | Complies |
| Microbiological Control | |||
| otal bacterial count | ≤1000cfu/g | USP/Ph.Eur | Complies |
| Yeast & mold | ≤100cfu/g | USP/Ph.Eur | Complies |
| Salmonella | Negative | USP/Ph.Eur | Complies |
| E.Coli | Negative | USP/Ph.Eur | Complies |
|
More information of TRB |
||
| Regulation certification | ||
| U.S.FDA,CEP,KOSHER HALAL GMP ISO Certificates | ||
| Reliable Quality | ||
| Nearly 20 years,export 40 countries and regions,more than 2000 batches produced by TRB have no any quality problems,unique purification process,impurity and purity control meet USP,EP and CP | ||
| Comprehensive Quality System | ||
|
|
▲Quality Assurance System |
√ |
| ▲ Document control |
√ |
|
| ▲ Validation System |
√ |
|
| ▲ Training System |
√ |
|
| ▲ Internal Audit Protocol |
√ |
|
| ▲ Suppler Audit System |
√ |
|
| ▲ Equipment Facilities System |
√ |
|
| ▲ Material Control System |
√ |
|
| ▲ Production Control System |
√ |
|
| ▲ Packaging Labeling System |
√ |
|
| ▲ Laboratory Control System |
√ |
|
| ▲ Verification Validation System |
√ |
|
| ▲ Regulatory Affairs System |
√ |
|
| Control Whole Sources and Processes | ||
| Strictly controlled all raw material,accessories and packaging materials.Preferred raw materials and accessories and packaging materials supplier with U.S. DMF number.Several raw material suppliers as supply assurance. | ||
| Strong Cooperative Institutions to support | ||
| Institute of botany/Institution of microbiology/Academy of Science and Technology/University | ||