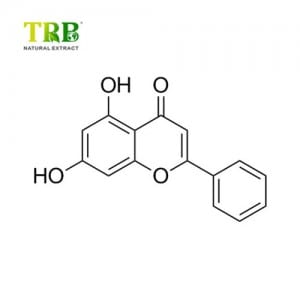
Product Name:Stevia Extract/Rebaudioside-A
Latin Name:Stevia rebaudiana (Bertoni) Hemsl
CAS No:57817-89-7;58543-16-1
Plant Part Used:Leaf
Assay:Stevioside; Rebaudioside A
Total Steviol Glycosides 98:Reb-A9≧97%, ≧98%, ≧99% by HPLC
Total Steviol Glycosides 95:Reb-A9≧50%, ≧60%, ≧80% by HPLC
Total Steviol Glycosides 90:Reb-A9≧40% by HPLC
Steviol Glycosides:90-95%;Stevioside 90-98%
Solubility:Soluble in water and ethanol
Colour:White powder with characteristic odor and taste
GMO Status:GMO Free
Packing: in 25kgs fiber drums
Storage:Keep container unopened in cool, dry place,Keep away from strong light
Shelf Life:24 months from date of production
Rebaudioside-A (Reb-A) Product Description
1. Product Overview
Rebaudioside-A (Reb-A) is a natural, high-intensity sweetener extracted from the leaves of the Stevia rebaudiana plant. With a sweetness 200-450 times that of sucrose and zero calories, it is an ideal sugar substitute for health-conscious consumers and food manufacturers seeking clean-label solutions . Approved by the FDA (2008) and EU (2011), Reb-A is widely used in beverages, dairy products, baked goods, and low-calorie jams .
2. Key Features
- Pure Sweetness: Reb-A delivers a clean, sugar-like taste without the bitter aftertaste common in other steviol glycosides .
- Heat Stability: Retains sweetness under high temperatures (up to 70°C), making it suitable for cooking and baking .
- Zero Calories & Blood Sugar Impact: Ideal for diabetics and weight management .
- Antioxidant & Antimicrobial Properties: Enhances product shelf life and nutritional value .
- Regulatory Compliance: Meets GRAS (Generally Recognized as Safe) standards and global food safety regulations .
3. Applications
- Beverages: Deep sugar reduction in soft drinks, teas, and functional beverages.
- Dairy & Desserts: Enhances sweetness in yogurts, ice creams, and sugar-free desserts.
- Confectionery: Used in candies, chewing gums, and low-calorie chocolate.
- Pharmaceuticals: Acts as a sweetening agent in syrups and chewable tablets.
Case Study: In sensory tests, a strawberry jam sweetened with 100% Reb-A outperformed sucralose in flavor and purchase intent, despite being 1.33 times less sweet than sucrose .
4. Technical Specifications
- Purity: ≥98% (HPLC grade) .
- Appearance: White to off-white crystalline powder .
- Solubility: Water-soluble, pH-stable in acidic conditions .
- Storage: Store in a cool, dry place (-20°C for long-term stability) .
5. Why Choose Reb-A?
- Consumer Preference: 54% of panelists favored Reb-A over sucralose in blind tests .
- Market Trends: Rising demand for natural, plant-based sweeteners in Europe and North America .
- Sustainability: Produced through eco-friendly enzymatic processes, reducing environmental impact .
6. Keywords
- “Natural Stevia Sweetener,” “Rebaudioside-A Supplier,” “Zero-Calorie Sweetener,” “FDA-Approved Stevia Extract.”
- “Reb-A for beverages,” “High-purity steviol glycosides,” “Non-GMO sugar substitute.”
- ”EU-certified,” “GRAS status,” and “vegan-friendly” to target regional preferences .
7. Compliance & Certifications
- FDA GRAS Notice No. GRN 000252 .
- EU Regulation (EC) No 1131/2011 .
- ISO 9001 & Halal/Kosher certifications available upon request.